Abstract
While allogeneic bone marrow transplant (alloBMT) can offer a curative approach for HIV, successes have been limited. HIV-specific CD8+ T cells may be harnessed to eliminate or control HIV, but these responses are limited by factors including viral immune escape mutations. In this study, we hypothesized that T cell therapies targeting conserved HIV epitopes would be safe and show in vivo antiviral activity. This phase I clinical trial (NCT03485963) evaluated the safety, immunologic and virologic responses of a novel HIV-1 multi-antigen specific T cell therapy (HST-NEET) targeting HIV Nef and conserved epitopes of Gag and Pol in people with HIV suppressed on antiretroviral therapy (ART). We investigated whether infused autologous HST-NEETs expand, persist, and elicit anti-viral responses in vivo.
Six participants with HIV (ages 33-57 years, 3 males and 2 females identifying as African or African American, 1 male identifying as White) received two infusions (2x107 cells/m2 BSA/dose) of HST-NEETs without prescribed lymphodepletion. Treatment follow-up was conducted for 48 weeks post-infusions. RESIST001 and 005 received infusions 4 weeks apart. RESIST003 and 004 received infusions >4 weeks apart (58 weeks and 35 weeks, respectively) due to delays caused by the COVID-19 pandemic. RESIST006 and 007 received infusions 2 weeks apart. RESIST004 was hospitalized for COVID-19 between infusions, but no attributable severe adverse events were observed for any participant irrespective of the timing of their infusions. All participants remained on ART throughout the study period.
To determine the epitope specificities and breadth of the T cell repertoire within the infusion products, IFNγ ELISPOT and intracellular cytokine flow cytometric assays were used to evaluate antigen-specific responses to HIV Gag, Pol, and Nef in the HST-NEET products. These studies also support tracking of the infused T cells in vivo. ELISPOT, flow cytometry and TCR sequencing were conducted to evaluate expansion and persistence of HIV-specific T cell clones in vivo. Pre- and post-infusion, the genetically intact HIV reservoir was quantified using the intact proviral DNA assay (IPDA), while HIV Envelope gp120 (Env)-specific antibody levels were assessed using ELISA.
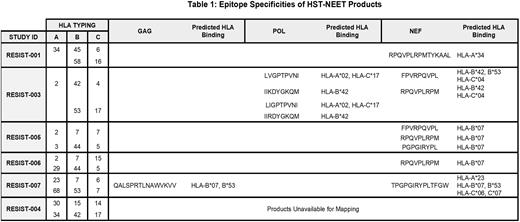
HST-NEET infusion products met clinical dose requirements, with predominantly CD3+CD8+ (med 70.7%, range 19% to 95.6%) populations expressing effector memory (CD45RO+CCR7-CD62L-) (86.8-98.4%) markers. HST-NEET products had specificity for 1-3 viral antigens with a dominance of Nef-specific responses (Table 1). Epitope mapping of the products identified 10 recognized epitopes, including repeated Nef epitopes associated with the HLA-B07 supertype.
Post-infusion studies (including TCR sequencing) for RESIST001 revealed marked in vivo expansion and persistence of T cells recognizing the same Nef peptide in the HST-NEETs product. However, no changes in HIV gp120 antibody levels or viral load were observed. RESIST005 HST-NEETs recognized 3 Nef epitopes but again, no changes in viral load were observed. RESIST004 also received a predominantly Nef-specific HST-NEET product eliciting expansion of Nef-specific T cells in vivo post-infusion. In contrast to RESIST001 and 005, a decrease in HIV reservoir size was observed in RESIST004 post-infusion #1 from 32.1 (pre) to 0 (at 36 weeks) intact proviruses/million CD4+ T cells (INTACT/M), which remained low at 8.4 INTACT/M post-infusion #2. RESIST003 products were broadly specific, mapping to 4 HIV Pol and 2 Nef CD8-restricted epitopes and at 5 weeks post-infusion #1, HIV reservoir size markedly decreased from 24.4 to 0 INTACT/M. Further, post HST-NEET infusion #2, the levels remained low (mean 6.3 INTACT/M). The immune and anti-viral studies are ongoing for RESIST006 and 007.
In summary, these data suggest that infusions of HST-NEETs are safe, can elicit expansion and persistence of HIV-specific T cell populations post-infusion even without prescribed lymphodepletion, and may be associated with durable decreases in the HIV reservoir, as measured by intact proviruses. Although these studies are ongoing, the infusion of HST-NEETs may be a viable therapeutic strategy in the lymphodepleted setting post autologous and allogeneic stem cell transplant as is also being evaluated in 2 ongoing clinical trials (NCT04975698 and NCT04248192, respectively).
Disclosures
Hanley:Mana Therapeutics: Current equity holder in private company, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Cellevolve: Consultancy, Other: Scientific Advisory Board; Cellenkos: Consultancy, Other: Scientific Advisory Board; MicroFluidx: Consultancy, Other: Scientific Advisory Board; Discovery Life Sciences: Consultancy, Other: Scientific Advisory Board. Cruz:Mana Therapeutics: Current equity holder in private company, Current holder of stock options in a privately-held company, Other: Co-Founder and Scientific Advisory Board , Patents & Royalties: HIV-specific T cells, antigen-specific T cells. Bollard:BMS: Consultancy; Pfizer: Consultancy; Roche: Consultancy; Mana Therapeutics: Current equity holder in private company, Current holder of stock options in a privately-held company, Other: scientific ci-founder and SAB member, Patents & Royalties: VSTs/TAA-T; Catamaran Bio: Current equity holder in private company, Current holder of stock options in a privately-held company, Other: scientific ci-founder and SAB member; Neximmune: Current equity holder in private company; Repertoire Immune Medicines: Current equity holder in private company; Cabalatte Bio: Membership on an entity's Board of Directors or advisory committees; Cellmedica: Patents & Royalties; SOBI: Other: DSMB member.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal